Results are sent solely to the doctor who requested the test 7 to 10 days after the LNS receives your blood sample. They are never sent directly to the patient.
In the event of a:
Positive result:
This means there is a high probability that the fetus has trisomy 21, 13 or 18. Nevertheless, NIPT is a screening test and the relevant professional bodies recommend having a positive NIPT result confirmed by way of an invasive diagnostic test (primarily an amniotic fluid test). Your gynaecologist will talk you through the results and next steps.
The cytogenetics team will analyze the sample of amniotic fluid taken and interpret the results. You may also be asked to attend a genetic consultation with our team.
In very rare cases, NIPT can detect other chromosomal anomalies (other trisomies, large duplication or deletion > 7Mb in size, or exceptionally chromosomal anomaly from maternal origin). Only chromosomal anomaly of medical interest for the ongoing pregnancy would be reported.
In case of detection of a rare chromosomal anomaly, medical genetic consultation is required to explain in details the anomaly, potential effects for the fetus (or for you), and need of further investigations.
Negative result:
This means that the probability of the fetus having trisomy 21, 13 or 18 is very low. In this case, your pregnancy will be monitored as planned.
Inconclusive result:
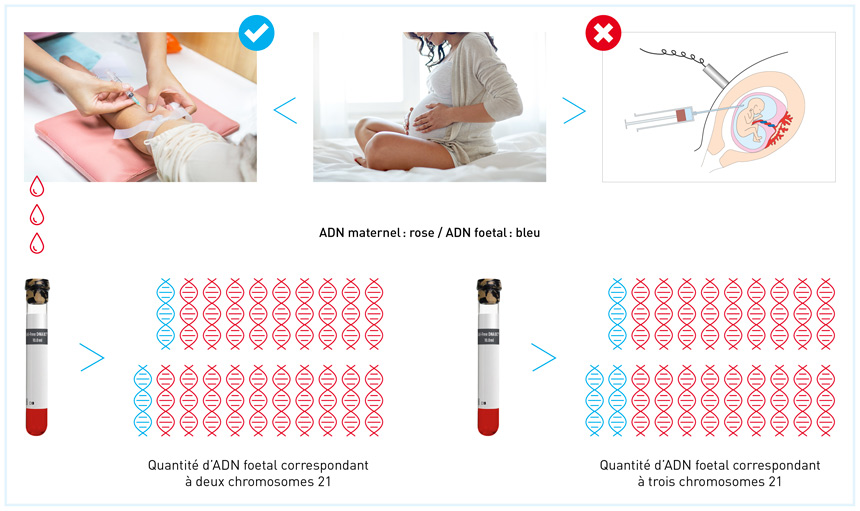
This means that it is not possible to reliably determine if the fetus has trisomy 13, 18 or 21. Inconclusive results are rare (around 1% of cases), but may occur for example when there is not enough cell-free fetal DNA (cffDNA) circulating in the blood sample taken from the mother. The proportion of cffDNA in the mother’s blood is known as the fetal fraction and is determined by many factors, including her BMI, if she is taking certain blood thinners, etc. Depending on your medical history, your gynaecologist may recommend you repeat the test or have further tests done.
Under no circumstances does having NIPT mean you do not have to go to your prenatal scans or attend check-ups with your obstetrician. This test is not a substitute for having your 1st trimester scan.